Trigger Point Therapy - Peripheral and Central Sensitization Explained
Trigger Points and Amplified Pain - Dr. Jonathan Kuttner
Working with Trigger Points - Where to Start?
Pain is a complex area of medicine, and current research has thrown up a number of discoveries relevant to trigger point manifestation and perpetuation
Pain systems need to be sensitive enough to detect potentially harmful stimuli. But in the case of trigger points, these systems eventually become too sensitive, causing us pain with no benefit.
Hypersensitivity arises because our pain pathways actually increase in sensitivity when they relay pain messages, and, with regard to MTPs, the mechanisms of this sensitization are now coming to light.
Peripheral Sensitization
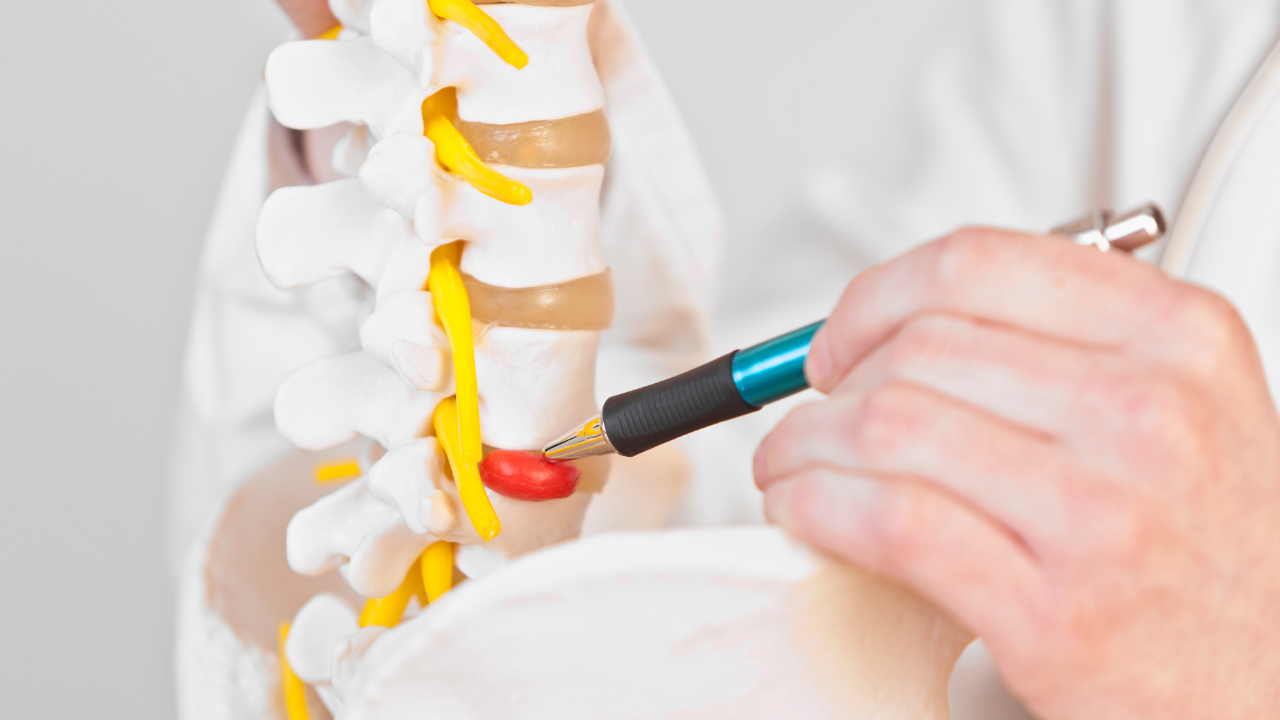
Within 48 hours of developing, and if untreated, MTPs cause inflammation, chronic facilitation, and changes in feedback from the host muscle.
Physiologically, there is a drop in the excitation threshold of polymodal nociceptors so that even normally innocuous, light stimuli activate them.
After sensitization of “pain fibers,” stimuli that as a rule are non-painful can cause pain and mechano-insensitive nerve fibers can become mechano-sensitive.
“This recruitment of silent nociceptors adds significantly to the nociceptive input to the spinal cord. Resting discharges may be induced or increased in nociceptors” (Schaible 2006).
This occurs because of chronic active trigger points providing a continuous afferent barrage into the spinal cord.
The suspected mechanism is:
-
Substance P, released from nociceptor terminals, carries nociceptive signals
for central processing, and alters local microcirculation and vessel permeability, leading to local edema, activating both mechanoreceptors and nociceptors, with subsequent increased tenderness and pain. - Persistent activation with these algogenic substances leads to changes in nociceptor responsiveness both peripherally and centrally.
It has been shown that up to 50% of muscle nerves may be made up of nociceptors, and that nociceptors also innervate the connective tissue surrounding muscle.
This could account for the severity of pain and exquisite tenderness found in muscles on palpation.
Persistent activation of nociceptors leads to peripheral sensitization whereby primary afferent nociceptors exhibit an enhanced responsiveness to natural stimuli.

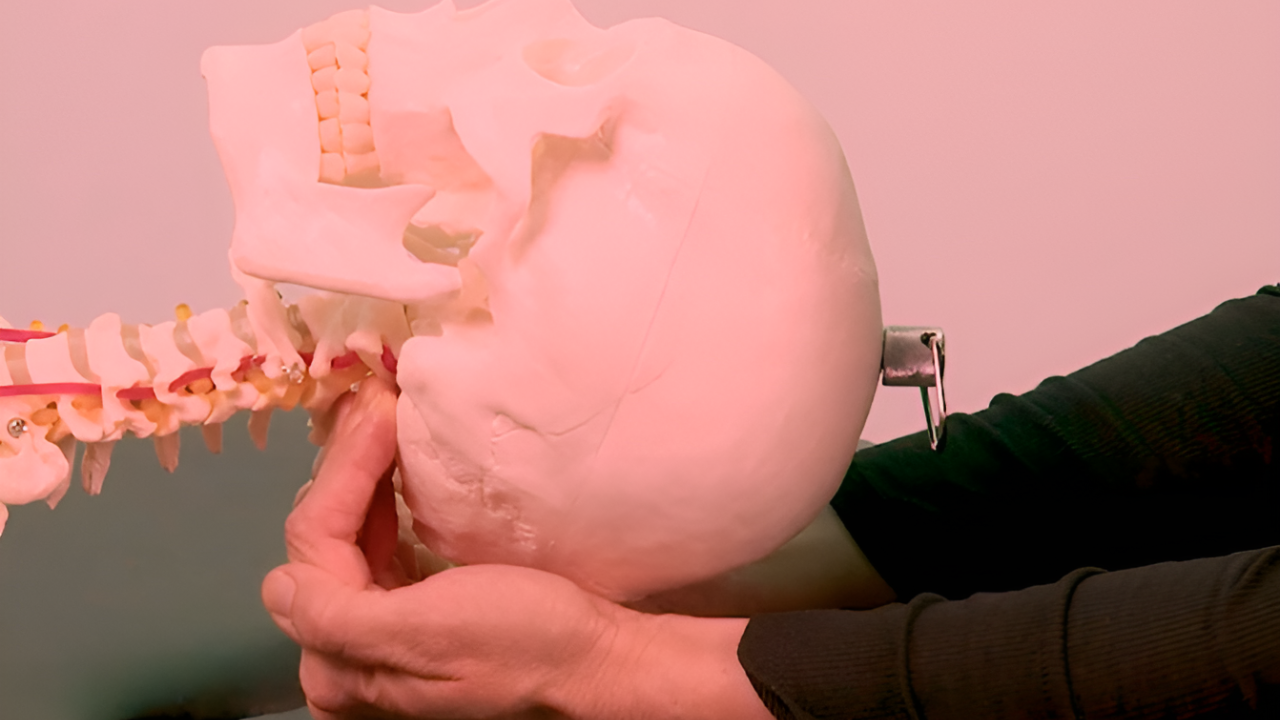
Central Sensitization (Spinal Hyperexcitability)
In the course of time the peripheral changes move deeper into the nervous system and the pattern becomes established centrally.
The superficial, the deep, and the ventral spinal cord show pronounced changes in their response properties (Schaible 2006). This is a form of neuroplasticity: after sensitization, an increased percentage of neurons in a segment respond to stimulation of an inflamed tissue.
The sensitivity of the spinal cord neurons becomes enhanced, so that an input that was previously subthreshold may now be sufficient to activate the neurons.
This effect is magnified up and down the spinal cord over several segmental levels both caudally and cephalically, which may lead to lowered activation thresholds for other MTPs.
The implications of this are profound: it may well be that a chronic trigger point in one area may sensitize levels of the spinal cord above and below the input level.
Over time, this may lead to a type of neuroplastic change in the CNS. This will decrease the pain threshold in other regions remote from the original source and possibly lower the threshold potential for other trigger points within the pain map.
Central sensitization can persist for weeks, months, and even years, depending on the chronicity of the stimulus.
The suspected mechanism is:
- Continuous activation of muscle nociceptors— induces neuroplastic changes and sensitization of dorsal horn neurons.
- Nociceptive input from skeletal muscle—far more effective at inducing neuroplastic change in the spinal cord than noxious input from the skin.
- Repetitive stimulation of primary afferent nociceptors leading to a progressive increase in action potential discharge—a phenomenon called windup, which may lead to a 20-fold increase in neuronal sensitivity.
- The result is an increase in intensity of pain and sensitization of neurones in the dorsal horn of the spinal cord because of the activation of N-methyl-D-aspartate (NMDA) receptors— central sensitization.
- Sensory neurones from the dorsal root ganglia become sensitized to mechanical stimuli, so that only mildly painful stimuli become more painful—mechanical hyperalgesia.
- Sustained nociceptive input from active trigger points may not only sensitize dorsal horn neurons, leading to hyperalgesia and allodynia, but also generate expanded referred pain regions.
Potential mechanisms for this phenomenon are the activation of previously redundant synapses at the dorsal horn, and the sprouting of new spinal terminals that broaden synaptic contacts at the dorsal horn, which may explain the referred pain seen with active trigger points.
Both peripheral and central sensitization can have serious unwanted effects. The advice therefore is to interfere with this process as soon as possible.
The good news is that myofascial trigger point release and dry needling techniques have both been reliably demonstrated to reduce these effects.
Find a Trigger Point Professional in your area
Dry Needling for Trigger Points
Certify as a Trigger Point Therapist
About NAT Courses:
As a manual therapist or exercise professional, there is only one way to expand your business - education!
Learning more skills increases the services that you offer and provides more opportunity for specialization.
Every NAT course is designed to build on what you already know, to empower you to treat more clients and grow your practice, with a minimal investment in time and money.
Recommended Starter Packs:
This trigger point therapy blog is intended to be used for information purposes only and is not intended to be used for medical diagnosis or treatment or to substitute for a medical diagnosis and/or treatment rendered or prescribed by a physician or competent healthcare professional. This information is designed as educational material, but should not be taken as a recommendation for treatment of any particular person or patient. Always consult your physician if you think you need treatment or if you feel unwell.

Learn More for Less
Unlimited access to all courses for just $19.95/mo